
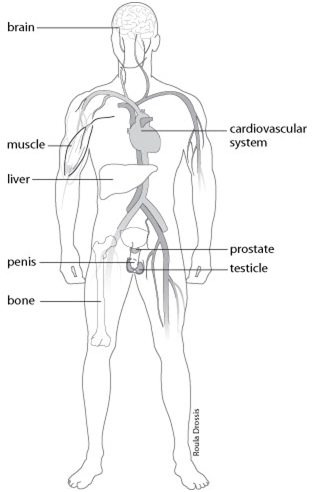
Subsequent advances included the identification, isolation, sequencing and regulatory characterization of the aromatase cytochrome P450 enzyme, product of the CYP19A1 gene, the enzyme responsible for aromatization of testosterone to 17β estradiol (E2) and androstenedione to estrone (E1), the major endogenous estrogens ( 4). In 1937 Steinach and Kun demonstrated that administration of large doses of testosterone to men increased the estrogenic activity of their urine and inferred in vivo conversion of testosterone to estrogens ( 3). Finally, E2 treatment, either as add-back to conventional ADT or as sole mode of ADT could be a useful strategy for men with prostate cancer.Įstrogens were demonstrated in the urine of men in the 1920s ( 1) and in the testis in 1952 ( 2). From a clinical perspective, the current evidence supports the use of testosterone as the treatment of choice in male hypogonadism, rather than aromatase inhibitors (which raise testosterone and lower E2), selective androgen receptor modulators and selective estrogen receptor modulators (with insufficiently understood tissue-specific estrogenic effects). In other tissues, particularly brain, elucidation of the clinical relevance of E2 actions requires further research. The collective evidence suggests that, in men, E2 is an important hormone for hypothalamic–pituitary–testicular axis regulation, reproductive function, growth hormone insulin-like growth factor-1 axis regulation, bone growth and maintenance of skeletal health, body composition and glucose metabolism and vasomotor stability. Human studies contributing evidence for the role of E2 in men comprise rare case reports of men lacking aromatase or a functional estrogen receptor alpha, short-term experiments manipulating sex steroid milieu in healthy men, men with organic hypogonadism or men with prostate cancer treated with androgen deprivation therapy (ADT) and from observational studies in community-dwelling men. In men, E2 circulates at concentrations exceeding those of postmenopausal women, and estrogen receptors are expressed in many male reproductive and somatic tissues. Evidence has been accumulating that, in men, some of the biological actions traditionally attributed to testosterone acting via the androgen receptor may in fact be dependent on its aromatization to estradiol (E2).
